- Home
- PHARMACEUTICAL BOTTLE
- Amber Moulded Vials for Injection
Amber Moulded Vials for Injection
- General Details
- Advantage
- Contact Us
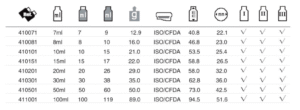
Classification of Moulded Glass Vial for Injection
Glass containers for pharmaceutical use are glass articles intended to come into direct contact with pharmaceutical preparations. The hydrolytic stability of glass containers for pharmaceutical use is expressed by the resistance to the release of soluble mineral substances into water under the prescribed conditions of contact between the inner surface of the container or glass grains and water. The hydrolytic resistance is evaluated by titrating released alkali. According to their hydrolytic resistance, glass containers are classified as follows:
| Glass Type | Hydrolytic resistance | Application |
| Type I | Neutral glass, a borosilicate glass containing significant amounts of boric oxide. With a high hydrolytic resistance due to the chemical composition of the glass itself. (moulded glass vial, and tubular vial) | Suitable for most preparations whether or not for parenteral use(injection use) |
| Type II | Soda-lime glass with sulfur treatment, with a high hydrolytic resistance resulting from suitable treatment of the surface of glass vial (only moulded glass vial) | Suitable for most acidic and neutral, aqueous preparations whether or not for parenteral use. |
| Type III | Soda-lime glass, with only moderate hydrolytic resistance (only moulded glass vial). Some factories use amber tubing glass container for oral use. | Generally suitable for non-aqueous preparations for parenteral use, for powders for parenteral use(not including freeze-dried preparations), and for preparations not for parenteral use. |
Shandong Pharmaceutical Packaging Group focuses on pharmaceutical packaging more than 21 years, and we are trying to be a top manufacturer of pharmaceutical packaging in China.
Our company realizes GMP clean production, complete product series, one-stop supply, and has become the Asia’s largest and one of the world top three suppliers of pharmaceutical glass packaging materials
1. Our company produces and supplies all pharmaceutical glass packaging materials under the norms of CFDA / ISO and the requirements specified in the current version of EP and USP.
2. Our products are exported to nearly 92 countries and regions, with the annual export scale of around 200 million USD.
3. It has successively passed relevant system certifications, including ISO15378, ISO9001, ISO14001, ISO45001, FSSC22000.
The pharmaceutical glass we produce almost covers the mainstream product types and specifications required in the market. At the same time, we also produce butyl rubber plugs for injection and related supporting caps.
Email: sales@pharmaceutical-glass.cn

